
Every six years, the Environment Agency publish data showing the state of our rivers relative to the standards required by the Water Framework Directive (which has been transcribed into UK law post-Brexit). This blog explores behind the headlines that caused outrage when the results were first published and led to a stinging blog by myself and reveals a very different picture to that which might be imagined if you only read headlines. In fact, the River Aire is improving – not a lot, but significantly.
My headline is not that all rivers fail, but that the Aire has improved in many ways. Whether it passes or fails depends upon what data you use and how you look at that data. It’s not a simple story, so settle back and read on…
I started work as a Pollution Prevention Officer on 1st July 1974 and have been involved, one way or another, with our river ever since and you might think, from the headlines, that all of the money, time and thought that has been put into improving the River Aire over the 45 years in which I have been involved has been wasted. That’s not true – so here is my interpretation of way over 250,000 individual ‘analyses’ done by the EA on the River Aire in the last three years. In order to do that, and to make what is happening understandable, I need to look at three key issues:
1) The ‘one out, all out’ rule
2) Ecological data
3) Chemical data
According to the ‘one out all out’ principle of the Water Framework Directive (WFD) good status of a water body is reached if all parameters (aka analyses) are classed as good. Missing one single parameter is sufficient to downgrade all of them. Essentially, if they analyse for 50 different chemicals and one fails then the whole river fails regardless of how good the other aspects of river chemistry are. Think about this. The more chemicals you analyse for, the more chance of the river failing regardless of whether there has actually been a deterioration and if you suddenly decide to analyse for a 51st chemical then the risk of failure increases even though the quality might not actually have changed. Well, this is what happened.
Whilst this might be considered as an application of the precautionary principle, the approach has several disadvantages. With such a principle, the classification obtained does not reflect any partial progress made, especially on the reduction of significant pressures. In some cases, parameters such as hydromorphological and physicochemical elements may better reflect changes in the water status while biological parameters are used to reflect the status of the whole ecosystem.
Moreover, it is subject to quirks such as introducing new parameters into the mix (such as Polybrominated Diphenyl Ethers – PBDEs, mentioned below) and thus increasing the chances of failure regardless of whether there has actually been any change. This is what happened to cause all waterbodies to fail for chemical status. See below for more detail.
Let’s look at the ecological data first – after all this is the good news relating to data about the fish, invertebrates, plants etc in the river. They just ‘sense’ the state of the river without bothering about pesky EU Directives – remember that we have salmon below Leeds waiting to find their way upstream to spawn and the middle/upper Aire is a strong trout and grayling fishery. They don’t do very well in failing rivers!
There is still some way to go, but steady progress is being made and many (most?) of the ‘Moderate’ classifications would be better were it not for the actions of our predecessor generations who thought they were doing right but weren’t. Many are Moderate because they have been designated as ‘Heavily Modified’ because they were canalised, culverted, had weirs installed, straightened, had stone or concrete walls put in place and so on, so they are no longer natural in appearance or function. Much of this damage to the natural state of our rivers will take generations to resolve and may never be done – how, for example, do you de-culvert Bradford Beck when it runs underneath some of the most iconic buildings in the centre of Bradford or the River Aire running beneath Leeds City railway station?
Now let’s look at the chemistry. The EA analyse for a very wide range of substances, from Dissolved Oxygen through Biochemical Oxygen Demand (the amount of degradable material in the water – often from sewage) to some real ‘exotics’ such as Polybrominated Diphenyl Ethers PBDEs – we will come back to these shortly. According to this data not one single waterbody passes! This is the data that caused all the fuss and it needs to be taken in context and the reasons for the mass failure understood.
Looking wider, the picture is actually improving, and faster than for the ecology. This chart shows the percentage of all the individual tests that passed the relevant limits for each of three years.
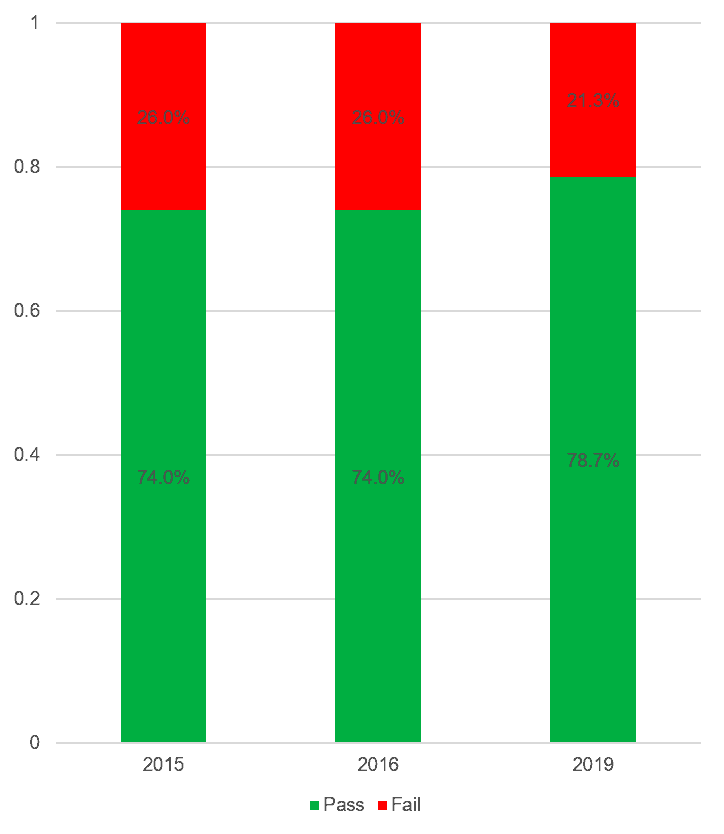
Again, there is still some way to go, but progress is being made.
Planned (and financed!) improvements to Phosphate and Ammonia discharges by Yorkshire Water will help substantially and we expect this to be significantly better in 5 years’ time.
So what happened to the chemistry of our rivers? Simple really – the EA started to analyse for a couple of exotic, yet ubiquitous, organic chemicals and found them everywhere (after all, they are ubiquitous!). PBDEs and something called Perfluorooctane Sulfonate (PFOS for short). They also changed their method for looking for Mercury and found it to be more widespread than first imagined.
Horror, you might think! Well, maybe not. These chemicals have been in our environment for decades,
PBDEs for instance have been used as flame retardants in a wide range of products including electrical and electronic equipment, textiles and foams. Releases occur during use and disposal of these products and they continue to enter waste water treatment works. They may be present in soil as a result of the spreading of sludge to land. PBDEs may also be released into the water column by the re-suspension of contaminated sediment or the transformation of decaBDE, which is still in use in industrial products. They may also volatilize and be deposited aerially.
PFOS has been used in a range of domestic consumer products, as well as in industrial processes and in aqueous film forming foams used in fire-fighting. Older consumer products such as carpets, textiles and upholstery that have been treated with PFOS or related substances will continue to act as a source of PFOS. Emissions can occur during the use, washing and disposal of such items, entering the environment via waste water treatment works, urban runoff or waste management facilities. PFOS can be present in soil from historical sources or from the spreading of sewage sludge to land.
And finally Mercury, which has been used in electrical equipment such as thermostats and batteries, cosmetics, wood preservatives, textile treatment agents, dental fillings, measurement devices and as an antifouling agent on boat hulls. A major use of mercury has been in mercury amalgam dental fillings, although this is now declining. Atmospheric releases of mercury are significantly greater than direct emissions to water, with thermal power plants and combustion installations being the largest industrial source emitters. Aerial deposition originating from outside the UK is now a significant source of mercury.
All three of these substances are now controlled (and substantially banned) internationally and are so ubiquitous and persistent that it is expected that they could be around for at least another decade. Realistically, there is little that a Rivers Trust or indeed the EA can do to further reduce these chemicals unless we find a point source discharge (we are checking, just in case).
If you want to look at the raw data yourself, you need to be looking here for chemical data and here for ecological data. Be warned – it’s a very clunky process and you will end up with huge quantitites of data in spreadsheet format. But if you persevere and are handy with Excel then it’s intriguing stuff.
This blog has opened the curtains and seen that things are better than might have been imagined. If you want to go a layer deeper in understanding the data and/or how the classification system works then do contact us.
Contrary to what the headlines might have suggested, the River Aire is actually improving for both chemical and ecological standards.
There is some way to go yet, but we have to be heartened by the fact that we have such a good existing fish population and by the plans of Yorkshire Water to spend large sums to further improve the situation.
Not that we will rest on any laurels – there is work to do and The Aire Rivers Trust plans to be at the forefront of pushing for and delivering the necessary improvements.
Hydromorphological?
Hydromorphology is a term used in river basin management to describe the hydrological (water flow, energy etc) and geomorphological (surface features) processes and attributes of rivers, lakes, estuaries and coastal waters. The Water Framework Directive (WFD) dictates that the ecology of surface waters is protected by correctly managing their hydrology and geomorphology.
Hydromorphology assessment and prediction requires an evaluation of aquatic habitat composition and the dynamic nature of water bodies and facilitates sustainable restoration.
Physicochemical?
The physicochemical status of a waterbody is linked to a series of measurments of the physical condition – Temperature for instance – and the basic chemistry – Ammonia, Phosphate, pH, BOD etc…
Dissolved Oxygen
This is the amount of oxygen dissolved in the water. All life needs oxygen, so this is an indicator of the ability to support life.
It is typically about 10 milligrams per litre, but the maiximum depends on the temperature of the water (the hotter it gets, the less oxygen the water can carry and so the more stressed the biota become).
Biochemical Oxygen Demand
Biochemical oxygen demand (BOD) represents the amount of oxygen consumed by bacteria and other microorganisms while they decompose organic matter under aerobic (oxygen is present) conditions at a specified temperature. The lower the better as high levels (above 5?) suggest pollution.

